Benzyl salicylate: Difference between revisions
Disambiguated: fixative → fixative (perfumery) |
Typo: its (possessive) not it's ("it is") |
||
| Line 37: | Line 37: | ||
}} |
}} |
||
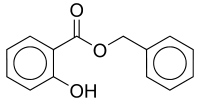
'''Benzyl salicylate''' is a [[salicylic acid]] benzyl [[ester]], a chemical compound most frequently used in cosmetics. It appears as an almost colorless liquid with a mild odor described as "very faint, sweet-floral, slightly balsamic" by those who can smell it, but many people either can't smell it at all or describe |
'''Benzyl salicylate''' is a [[salicylic acid]] benzyl [[ester]], a chemical compound most frequently used in cosmetics. It appears as an almost colorless liquid with a mild odor described as "very faint, sweet-floral, slightly balsamic" by those who can smell it, but many people either can't smell it at all or describe its smell as "musky". Trace impurities can have a significant influence on the odour.<ref>Steffen Arctander: Perfume and Flavor Chemicals. ISBN 0-931710-37-5</ref> It occurs naturally in a variety of plants and plant extracts and is widely used in blends of fragrance materials.<ref name=thegoodscentscompany>{{cite web | url = http://www.thegoodscentscompany.com/data/rw1001791.html | title = Benzyl salicylate | publisher = The Good Scents Company}}</ref> |
||
There is some evidence that people can become sensitized to this material<ref>{{cite journal | url = http://www.rifm.org/doc/Food%20&%20Chem%20Tox%20RIFM%20Spec%20Suppl%20122007.pdf | journal = Food and Chemical Technology | volume = 45 | issue = Supplement 1 | year = 2007 | title = Toxicologic and Dermatologic Assessments for Three Groups of Fragrance Ingredients: 1) Related Esters and Alcohols of Cinnamic Acid and Cinnamic Alcohol, 2) Ionones, 3) Salicylates}}</ref> and as a result there is a restriction standard concerning the use of this material in fragrances by the [[International Fragrance Association]].<ref>{{cite web | url = http://www.ifraorg.org/en-us/standards_restricted/s3/p2 | title = Standards Restricted | publisher = [[International Fragrance Association]]}}</ref> |
There is some evidence that people can become sensitized to this material<ref>{{cite journal | url = http://www.rifm.org/doc/Food%20&%20Chem%20Tox%20RIFM%20Spec%20Suppl%20122007.pdf | journal = Food and Chemical Technology | volume = 45 | issue = Supplement 1 | year = 2007 | title = Toxicologic and Dermatologic Assessments for Three Groups of Fragrance Ingredients: 1) Related Esters and Alcohols of Cinnamic Acid and Cinnamic Alcohol, 2) Ionones, 3) Salicylates}}</ref> and as a result there is a restriction standard concerning the use of this material in fragrances by the [[International Fragrance Association]].<ref>{{cite web | url = http://www.ifraorg.org/en-us/standards_restricted/s3/p2 | title = Standards Restricted | publisher = [[International Fragrance Association]]}}</ref> |
||
Revision as of 15:55, 23 June 2013
| |
| Names | |
|---|---|
| IUPAC name
Benzyl 2-hydroxybenzoate
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.003.876 |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C14H12O3 | |
| Molar mass | 228.247 g·mol−1 |
| Appearance | Colorless liquid |
| Density | 1.17 g/cm3 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Benzyl salicylate is a salicylic acid benzyl ester, a chemical compound most frequently used in cosmetics. It appears as an almost colorless liquid with a mild odor described as "very faint, sweet-floral, slightly balsamic" by those who can smell it, but many people either can't smell it at all or describe its smell as "musky". Trace impurities can have a significant influence on the odour.[1] It occurs naturally in a variety of plants and plant extracts and is widely used in blends of fragrance materials.[2]
There is some evidence that people can become sensitized to this material[3] and as a result there is a restriction standard concerning the use of this material in fragrances by the International Fragrance Association.[4]
It is used as a solvent for crystalline synthetic musks and as a component and fixative in floral perfumes such as carnation, jasmine, lilac, and wallflower.[5]
References
- ^ Steffen Arctander: Perfume and Flavor Chemicals. ISBN 0-931710-37-5
- ^ "Benzyl salicylate". The Good Scents Company.
- ^ "Toxicologic and Dermatologic Assessments for Three Groups of Fragrance Ingredients: 1) Related Esters and Alcohols of Cinnamic Acid and Cinnamic Alcohol, 2) Ionones, 3) Salicylates" (PDF). Food and Chemical Technology. 45 (Supplement 1). 2007.
- ^ "Standards Restricted". International Fragrance Association.
- ^ An Introduction to Perfumery by Curtis & Williams 2nd Edition, 2009, ISBN 978-0-9608752-8-3, ISBN 978-1-870228-24-4
